Battery Cathodes: Intercalation Potential and Volume Change¶
Use the UMA-S-1.2-OMat machine-learning potential to optimize olivine LiFePO4 and FePO4, then estimate the average Li intercalation potential and the relative volume change on delithiation.
Battery Cathodes: Intercalation Potential and Volume Change¶
Cathode materials are often judged by more than their voltage alone. A useful battery cathode should store lithium at a favorable potential, but it should also remain structurally stable during charge and discharge. Large changes in unit-cell volume can lead to mechanical stress, cracking, and capacity fade over repeated cycling.
In this notebook we use the inorganic-materials UMA model, UMA-S-1.2-OMat, to optimize olivine LiFePO4 and its delithiated FePO4 counterpart. From these calculations we estimate two useful screening descriptors: the average lithium intercalation potential and the relative volume change upon delithiation.
Overview¶
The workflow follows the reaction
and evaluates it with a machine-learning potential. We will:
optimize bulk Li to obtain a consistent Li metal reference energy
optimize lithiated LiFePO4 and delithiated FePO4
compare the optimized structures visually and through their cell volumes
compute the average intercalation potential from the total energies
compute the relative volume change per formula unit and compare with experiment
from math import gcd
from ase.build import bulk
from scm.plams import AMSJob, Settings, Units, fromASE, view, plot_image_grid
Settings and Visualization Helpers¶
We use the UMA-S-1.2-OMat machine-learning potential with a geometry optimization and lattice relaxation so that both atomic positions and unit-cell parameters can respond to lithiation state. The D4 dispersion add-on is also consistently included in all calculations.
This section also defines a few small helper functions for structure handling, formula counting, and visualization.
def umat_settings(nproc=1):
s = Settings()
s.input.ams.Task = "GeometryOptimization"
s.input.ams.GeometryOptimization.OptimizeLattice = "Yes"
s.input.ams.EngineAddons.D4Dispersion.Enabled = "Yes"
s.input.mlpotential.Model = "UMA-S-1.2-OMat"
s.runscript.nproc = nproc
s.runscript.preamble_lines = ["export OMP_NUM_THREADS=1"]
return s
def formula_units(system):
counts = [count for count in system.get_formula(as_dict=True).values() if count > 0]
nfu = counts[0]
for count in counts[1:]:
nfu = gcd(nfu, count)
return nfu
def render(system, supercell=(3, 3, 1), direction="along_y", width=400, height=400, padding=-1):
m = system.copy().supercell(*supercell)
return view(
m,
direction=direction,
width=width,
height=height,
padding=padding,
fixed_atom_size=False,
show_atom_labels=False,
)
def make_lifepo4():
mol = AMSJob.from_input(
"""
System
Atoms
Li 0.0 0.0 0.0
Li 2.327458595 2.98537755 5.118098025
Li 2.327458595 0.0 5.118098025
Li 0.0 2.98537755 0.0
Fe 2.466481095 1.492688775 7.9960175444
Fe 0.1390225 4.478066325 7.3582765306
Fe 4.51589469 1.492688775 2.8779195194
Fe 2.188436095 4.478066325 2.2401785056
P 1.9486533972 1.492688775 0.9608338493
P 4.2761119922 4.478066325 4.1572641757
P 0.3788051978 1.492688775 6.0789318743
P 2.7062637928 4.478066325 9.2753622007
O 3.466919761 1.492688775 0.964563612
O 3.3223789033 3.25739339 8.5385692027
O 3.3223789033 5.69873926 8.5385692027
O 1.187997429 4.478066325 9.271632437999999
O 3.3043602309 4.478066325 0.4535511069
O 1.139461166 4.478066325 4.153534413
O 1.3325382867 0.27201584 1.6976268473
O 0.9949203083 2.71336171 6.8157248723
O 3.6599968817 3.25739339 3.4204711777
O 3.6599968817 5.69873926 3.4204711777
O 1.3505569591 1.492688775 9.782644943099999
O 0.9769016359 1.492688775 4.6645469181
O 3.6780155541 4.478066325 5.5716491319
O 3.515456024 1.492688775 6.082661637
O 0.9949203083 0.27201584 6.8157248723
O 1.3325382867 2.71336171 1.6976268473
End
Lattice
4.65491719 0.0 0.0
0.0 5.9707551 0.0
0.0 0.0 10.23619605
End
End
"""
).molecule
if isinstance(mol, dict) and len(mol) == 1 and "" in mol:
return mol[""]
else:
return mol
Lithium Metal Reference¶
To convert total-energy differences into an intercalation voltage, we need a reference for metallic lithium. We therefore optimize bulk bcc Li and extract the energy per Li atom.
li_bulk = fromASE(bulk("Li", "bcc", a=3.49, cubic=True))
li_job = AMSJob(name="Li_bcc_opt", molecule=li_bulk, settings=umat_settings())
li_result = li_job.run()
li_bulk_opt = li_result.get_main_molecule()
n_li = li_bulk_opt.get_formula(as_dict=True)["Li"]
e_li_metal = li_result.get_energy(unit="hartree") / n_li
v_li_bulk = li_bulk_opt.unit_cell_volume()
print(f"Li metal reference energy: {e_li_metal:.8f} Ha/atom")
print(f"Li bcc cell volume: {v_li_bulk:.5f} A^3")
[24.04|10:44:05] JOB Li_bcc_opt STARTED
[24.04|10:44:05] JOB Li_bcc_opt RUNNING
[24.04|10:44:29] JOB Li_bcc_opt FINISHED
[24.04|10:44:29] JOB Li_bcc_opt SUCCESSFUL
Li metal reference energy: -0.07082951 Ha/atom
Li bcc cell volume: 40.53114 A^3
plot_image_grid(
{"optimized Li metal": render(li_bulk_opt, direction="along_c", width=420, height=420, padding=0)},
figsize=(4, 4)
);
[24.04|10:44:32] Starting Xvfb...
[24.04|10:44:32] Xvfb started

Lithiated LiFePO4 Optimization¶
We define the olivine LiFePO4 unit cell directly in the notebook and optimize the fully lithiated cathode. The resulting total energy is one of the three quantities needed for the voltage calculation, and the relaxed cell volume provides the reference volume for the charged-state comparison.
lfp = make_lifepo4()
print(lfp.get_formula(False))
Fe4Li4O16P4
lfp_job = AMSJob(name="LiFePO4_opt", molecule=lfp, settings=umat_settings())
lfp_result = lfp_job.run()
lfp_opt = lfp_result.get_main_molecule()
e_lfp = lfp_result.get_energy(unit="hartree")
v_lfp = lfp_opt.unit_cell_volume()
print(f"LiFePO4 total energy: {e_lfp:.8f} Ha")
print(f"LiFePO4 cell volume: {v_lfp:.5f} A^3")
print(lfp_opt.get_formula(False))
[24.04|10:44:33] JOB LiFePO4_opt STARTED
[24.04|10:44:33] JOB LiFePO4_opt RUNNING
[24.04|10:52:38] JOB LiFePO4_opt FINISHED
[24.04|10:52:38] JOB LiFePO4_opt SUCCESSFUL
LiFePO4 total energy: -7.21135335 Ha
LiFePO4 cell volume: 293.76580 A^3
Fe4Li4O16P4
plot_image_grid({
"initial LiFePO4": render(lfp),
"optimized LiFePO4": render(lfp_opt),
});
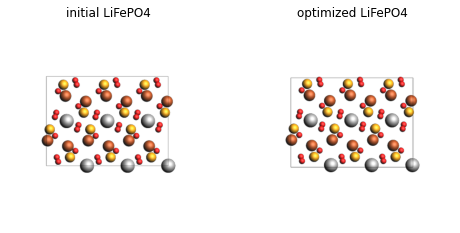
Delithiated FePO4¶
To represent delithiation, we remove all Li atoms from the optimized LiFePO4 structure and use the resulting FePO4 framework as the starting point for a second optimization.
This mirrors the structural change associated with charge extraction and lets us compare the two end members on the same footing.
fp = lfp_opt.copy()
for atom in list(fp):
if atom.symbol == "Li":
fp.delete_atom(atom)
print(fp.get_formula(False))
Fe4O16P4
fp_job = AMSJob(name="FePO4_opt", molecule=fp, settings=umat_settings())
fp_result = fp_job.run()
fp_opt = fp_result.get_main_molecule()
e_fp = fp_result.get_energy(unit="hartree")
v_fp = fp_opt.unit_cell_volume()
print(f"FePO4 total energy: {e_fp:.8f} Ha")
print(f"FePO4 cell volume: {v_fp:.5f} A^3")
print(fp_opt.get_formula(False))
[24.04|10:52:40] JOB FePO4_opt STARTED
[24.04|10:52:40] JOB FePO4_opt RUNNING
[24.04|11:00:24] JOB FePO4_opt FINISHED
[24.04|11:00:24] JOB FePO4_opt SUCCESSFUL
FePO4 total energy: -6.40276126 Ha
FePO4 cell volume: 276.83401 A^3
Fe4O16P4
plot_image_grid({
"optimized LiFePO4": render(lfp_opt),
"optimized FePO4": render(fp_opt),
});
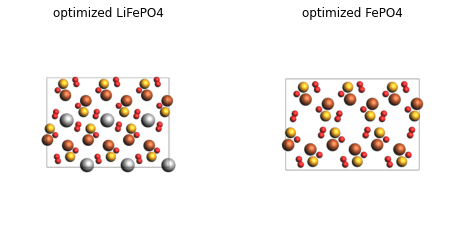
Analysis¶
With optimized energies and cell volumes for Li, LiFePO4, and FePO4 in hand, we can evaluate two useful cathode descriptors.
The average intercalation potential is obtained from the reaction energy, while the relative volume change measures how strongly the crystal expands or contracts on delithiation. For meaningful comparison across cells, both quantities are reduced to a per-formula-unit basis where needed.
n_formula_units = formula_units(lfp_opt)
e_lfp_fu = e_lfp / n_formula_units
e_fp_fu = e_fp / n_formula_units
v_lfp_fu = v_lfp / n_formula_units
v_fp_fu = v_fp / n_formula_units
voltage = Units.convert(e_fp_fu + e_li_metal - e_lfp_fu, "hartree", "eV")
delta_v_percent = 100.0 * (v_fp_fu - v_lfp_fu) / v_lfp_fu
print(f"LiFePO4 energy per formula unit: {e_lfp_fu:.8f} Ha")
print(f"FePO4 energy per formula unit: {e_fp_fu:.8f} Ha")
print(f"LiFePO4 volume per formula unit: {v_lfp_fu:.5f} A^3")
print(f"FePO4 volume per formula unit: {v_fp_fu:.5f} A^3")
print(f"Average intercalation potential: {voltage:.3f} V")
print(f"Relative volume change: {delta_v_percent:.3f} %")
LiFePO4 energy per formula unit: -1.80283834 Ha
FePO4 energy per formula unit: -1.60069031 Ha
LiFePO4 volume per formula unit: 73.44145 A^3
FePO4 volume per formula unit: 69.20850 A^3
Average intercalation potential: 3.573 V
Relative volume change: -5.764 %
Comparison to Experiment¶
A quick comparison to representative literature values helps place the calculation in context. Agreement at this level is useful as a sanity check before extending the same workflow to doped olivines, sodium analogues, or larger screening studies.
import pandas as pd
experimental = {
"LiFePO4 volume (A^3/f.u.)": 72.85,
"FePO4 volume (A^3/f.u.)": 68.09,
"Volume change (%)": -6.534,
"Intercalation potential (V)": 3.5,
}
calculated = {
"LiFePO4 volume (A^3/f.u.)": v_lfp_fu,
"FePO4 volume (A^3/f.u.)": v_fp_fu,
"Volume change (%)": delta_v_percent,
"Intercalation potential (V)": voltage,
}
df = pd.DataFrame([experimental, calculated], index=["Experimental", "Calculated"])
df.loc["Delta"] = df.loc["Calculated"] - df.loc["Experimental"]
df = df.round(3)
df
LiFePO4 volume (A^3/f.u.) | FePO4 volume (A^3/f.u.) | Volume change (%) | Intercalation potential (V) | |
|---|---|---|---|---|
Experimental | 72.850 | 68.090 | -6.534 | 3.500 |
Calculated | 73.441 | 69.209 | -5.764 | 3.573 |
Delta | 0.591 | 1.119 | 0.770 | 0.073 |
Extensions¶
The same workflow can be generalized in several directions:
replace Fe with a dopant and examine the trade-off between voltage and structural change
adapt the workflow to a sodium analogue and compare Na insertion with Li insertion
loop over multiple cathode materials and collect a benchmark table automatically
See also¶
References¶
Python Script¶
#!/usr/bin/env python
# coding: utf-8
# ## Battery Cathodes: Intercalation Potential and Volume Change
#
# Cathode materials are often judged by more than their voltage alone. A useful battery cathode should store lithium at a favorable potential, but it should also remain structurally stable during charge and discharge. Large changes in unit-cell volume can lead to mechanical stress, cracking, and capacity fade over repeated cycling.
#
# In this notebook we use the inorganic-materials UMA model, UMA-S-1.2-OMat, to optimize olivine LiFePO4 and its delithiated FePO4 counterpart. From these calculations we estimate two useful screening descriptors: the average lithium intercalation potential and the relative volume change upon delithiation.
# ## Overview
#
# The workflow follows the reaction
#
# $$\mathrm{LiFePO_4 \rightarrow FePO_4 + Li}$$
#
# and evaluates it with a machine-learning potential. We will:
#
# - optimize bulk Li to obtain a consistent Li metal reference energy
# - optimize lithiated LiFePO4 and delithiated FePO4
# - compare the optimized structures visually and through their cell volumes
# - compute the average intercalation potential from the total energies
# - compute the relative volume change per formula unit and compare with experiment
from math import gcd
from ase.build import bulk
from scm.plams import AMSJob, Settings, Units, fromASE, view, plot_image_grid
# ## Settings and Visualization Helpers
#
# We use the UMA-S-1.2-OMat machine-learning potential with a geometry optimization and lattice relaxation so that both atomic positions and unit-cell parameters can respond to lithiation state. The D4 dispersion add-on is also consistently included in all calculations.
#
# This section also defines a few small helper functions for structure handling, formula counting, and visualization.
def umat_settings(nproc=1):
s = Settings()
s.input.ams.Task = "GeometryOptimization"
s.input.ams.GeometryOptimization.OptimizeLattice = "Yes"
s.input.ams.EngineAddons.D4Dispersion.Enabled = "Yes"
s.input.mlpotential.Model = "UMA-S-1.2-OMat"
s.runscript.nproc = nproc
s.runscript.preamble_lines = ["export OMP_NUM_THREADS=1"]
return s
def formula_units(system):
counts = [count for count in system.get_formula(as_dict=True).values() if count > 0]
nfu = counts[0]
for count in counts[1:]:
nfu = gcd(nfu, count)
return nfu
def render(system, supercell=(3, 3, 1), direction="along_y", width=400, height=400, padding=-1):
m = system.copy().supercell(*supercell)
return view(
m,
direction=direction,
width=width,
height=height,
padding=padding,
fixed_atom_size=False,
show_atom_labels=False,
)
def make_lifepo4():
mol = AMSJob.from_input(
"""
System
Atoms
Li 0.0 0.0 0.0
Li 2.327458595 2.98537755 5.118098025
Li 2.327458595 0.0 5.118098025
Li 0.0 2.98537755 0.0
Fe 2.466481095 1.492688775 7.9960175444
Fe 0.1390225 4.478066325 7.3582765306
Fe 4.51589469 1.492688775 2.8779195194
Fe 2.188436095 4.478066325 2.2401785056
P 1.9486533972 1.492688775 0.9608338493
P 4.2761119922 4.478066325 4.1572641757
P 0.3788051978 1.492688775 6.0789318743
P 2.7062637928 4.478066325 9.2753622007
O 3.466919761 1.492688775 0.964563612
O 3.3223789033 3.25739339 8.5385692027
O 3.3223789033 5.69873926 8.5385692027
O 1.187997429 4.478066325 9.271632437999999
O 3.3043602309 4.478066325 0.4535511069
O 1.139461166 4.478066325 4.153534413
O 1.3325382867 0.27201584 1.6976268473
O 0.9949203083 2.71336171 6.8157248723
O 3.6599968817 3.25739339 3.4204711777
O 3.6599968817 5.69873926 3.4204711777
O 1.3505569591 1.492688775 9.782644943099999
O 0.9769016359 1.492688775 4.6645469181
O 3.6780155541 4.478066325 5.5716491319
O 3.515456024 1.492688775 6.082661637
O 0.9949203083 0.27201584 6.8157248723
O 1.3325382867 2.71336171 1.6976268473
End
Lattice
4.65491719 0.0 0.0
0.0 5.9707551 0.0
0.0 0.0 10.23619605
End
End
"""
).molecule
if isinstance(mol, dict) and len(mol) == 1 and "" in mol:
return mol[""]
else:
return mol
# ## Lithium Metal Reference
#
# To convert total-energy differences into an intercalation voltage, we need a reference for metallic lithium. We therefore optimize bulk bcc Li and extract the energy per Li atom.
li_bulk = fromASE(bulk("Li", "bcc", a=3.49, cubic=True))
li_job = AMSJob(name="Li_bcc_opt", molecule=li_bulk, settings=umat_settings())
li_result = li_job.run()
li_bulk_opt = li_result.get_main_molecule()
n_li = li_bulk_opt.get_formula(as_dict=True)["Li"]
e_li_metal = li_result.get_energy(unit="hartree") / n_li
v_li_bulk = li_bulk_opt.unit_cell_volume()
print(f"Li metal reference energy: {e_li_metal:.8f} Ha/atom")
print(f"Li bcc cell volume: {v_li_bulk:.5f} A^3")
plot_image_grid(
{"optimized Li metal": render(li_bulk_opt, direction="along_c", width=420, height=420, padding=0)},
figsize=(4, 4)
, save_path="picture1.png");
# ## Lithiated LiFePO4 Optimization
#
# We define the olivine LiFePO4 unit cell directly in the notebook and optimize the fully lithiated cathode. The resulting total energy is one of the three quantities needed for the voltage calculation, and the relaxed cell volume provides the reference volume for the charged-state comparison.
lfp = make_lifepo4()
print(lfp.get_formula(False))
lfp_job = AMSJob(name="LiFePO4_opt", molecule=lfp, settings=umat_settings())
lfp_result = lfp_job.run()
lfp_opt = lfp_result.get_main_molecule()
e_lfp = lfp_result.get_energy(unit="hartree")
v_lfp = lfp_opt.unit_cell_volume()
print(f"LiFePO4 total energy: {e_lfp:.8f} Ha")
print(f"LiFePO4 cell volume: {v_lfp:.5f} A^3")
print(lfp_opt.get_formula(False))
plot_image_grid({
"initial LiFePO4": render(lfp),
"optimized LiFePO4": render(lfp_opt),
}, save_path="picture2.png");
# ## Delithiated FePO4
#
# To represent delithiation, we remove all Li atoms from the optimized LiFePO4 structure and use the resulting FePO4 framework as the starting point for a second optimization.
#
# This mirrors the structural change associated with charge extraction and lets us compare the two end members on the same footing.
fp = lfp_opt.copy()
for atom in list(fp):
if atom.symbol == "Li":
fp.delete_atom(atom)
print(fp.get_formula(False))
fp_job = AMSJob(name="FePO4_opt", molecule=fp, settings=umat_settings())
fp_result = fp_job.run()
fp_opt = fp_result.get_main_molecule()
e_fp = fp_result.get_energy(unit="hartree")
v_fp = fp_opt.unit_cell_volume()
print(f"FePO4 total energy: {e_fp:.8f} Ha")
print(f"FePO4 cell volume: {v_fp:.5f} A^3")
print(fp_opt.get_formula(False))
plot_image_grid({
"optimized LiFePO4": render(lfp_opt),
"optimized FePO4": render(fp_opt),
}, save_path="picture3.png");
# ## Analysis
#
# With optimized energies and cell volumes for Li, LiFePO4, and FePO4 in hand, we can evaluate two useful cathode descriptors.
#
# The average intercalation potential is obtained from the reaction energy, while the relative volume change measures how strongly the crystal expands or contracts on delithiation. For meaningful comparison across cells, both quantities are reduced to a per-formula-unit basis where needed.
n_formula_units = formula_units(lfp_opt)
e_lfp_fu = e_lfp / n_formula_units
e_fp_fu = e_fp / n_formula_units
v_lfp_fu = v_lfp / n_formula_units
v_fp_fu = v_fp / n_formula_units
voltage = Units.convert(e_fp_fu + e_li_metal - e_lfp_fu, "hartree", "eV")
delta_v_percent = 100.0 * (v_fp_fu - v_lfp_fu) / v_lfp_fu
print(f"LiFePO4 energy per formula unit: {e_lfp_fu:.8f} Ha")
print(f"FePO4 energy per formula unit: {e_fp_fu:.8f} Ha")
print(f"LiFePO4 volume per formula unit: {v_lfp_fu:.5f} A^3")
print(f"FePO4 volume per formula unit: {v_fp_fu:.5f} A^3")
print(f"Average intercalation potential: {voltage:.3f} V")
print(f"Relative volume change: {delta_v_percent:.3f} %")
# ## Comparison to Experiment
#
# A quick comparison to representative literature values helps place the calculation in context. Agreement at this level is useful as a sanity check before extending the same workflow to doped olivines, sodium analogues, or larger screening studies.
import pandas as pd
experimental = {
"LiFePO4 volume (A^3/f.u.)": 72.85,
"FePO4 volume (A^3/f.u.)": 68.09,
"Volume change (%)": -6.534,
"Intercalation potential (V)": 3.5,
}
calculated = {
"LiFePO4 volume (A^3/f.u.)": v_lfp_fu,
"FePO4 volume (A^3/f.u.)": v_fp_fu,
"Volume change (%)": delta_v_percent,
"Intercalation potential (V)": voltage,
}
df = pd.DataFrame([experimental, calculated], index=["Experimental", "Calculated"])
df.loc["Delta"] = df.loc["Calculated"] - df.loc["Experimental"]
df = df.round(3)
print(df)
# ## Extensions
#
# The same workflow can be generalized in several directions:
#
# - replace Fe with a dopant and examine the trade-off between voltage and structural change
# - adapt the workflow to a sodium analogue and compare Na insertion with Li insertion
# - loop over multiple cathode materials and collect a benchmark table automatically