Polymers¶
The COSMO-RS program can also be used to calculate thermodynamic properties of polymers and polymer-containing mixtures. The theory and equations behind this are described in the COSMO-RS documentation. This tutorial will focus on practical details of performing calculations with polymers in the COSMO-RS GUI.
This tutorial expects that that you are familiar with setting COSMO-RS parameters and pure compound data (see the COSMO-RS overview tutorial: parameters and analysis), doing COSMO-RS calculations (see the COSMO-RS overview tutorial: properties), and that the COSMO-RS compound database ADFCRS-2018 has been installed already (see the COSMO-RS compound database tutorial).
The ADFCRS Polymer Database¶
The COSMO-RS Polymer Database contains >200 structures of common polymers, all represented as the central unit of a trimer as described in the COSMO-RS manual. Because the simplest procedure used to generate the structure of a monomer still requires the calculation of a trimer, it is recommended to use the polymer database for this tutorial as adding user-defined polymers will likely be time-consuming (minutes-days, depending on the size of the polymer).
To quickly get started with polymers in COSMO-RS, download the COSMO-RS Polymer Database. More information about the database can be found here.
Selecting/inputting database compounds¶
After downloading and unzipping the database, open the COSMO-RS GUI AMScrs. To use the database in COSMO-RS, follow the steps shown below.
Note
Polymeric compounds can also be added by SMILES string. Be sure to input a polymer in CurlySMILES format. One can read the CurlySMILES paper for more thorough coverage, but the CurlySMILES format is fairly self-explanatory. For example, in CurlySMILES polyethylene is C{-}C{n+} and polysytrene is C{-}C{n+}(c1ccccc1). These examples show that the atoms that are part of the polymerization are marked with {-} and {n+} identifiers.
Inputting your own polymers (optional)¶
It is also possible to input new polymers to the database and use these structures as you would any polymer in the database. As for all new compound inputs, there are two ways of doing this.
Method 1: Using ADF¶
This is the standard method for using compounds not already present in the database. This method requires an ADF and COSMO calculation, but polymer calculations can require a lot of atoms, sometimes making these calculations expensive. For this method, we must first perform a geometry optimization and then a single point COSMO calculation. The required steps for this calculation are as follows:
We will illustrate the process of new polymer input using polyethylene below:
(1) Select Task → COSMO-RS Compound, Input the monomer structure
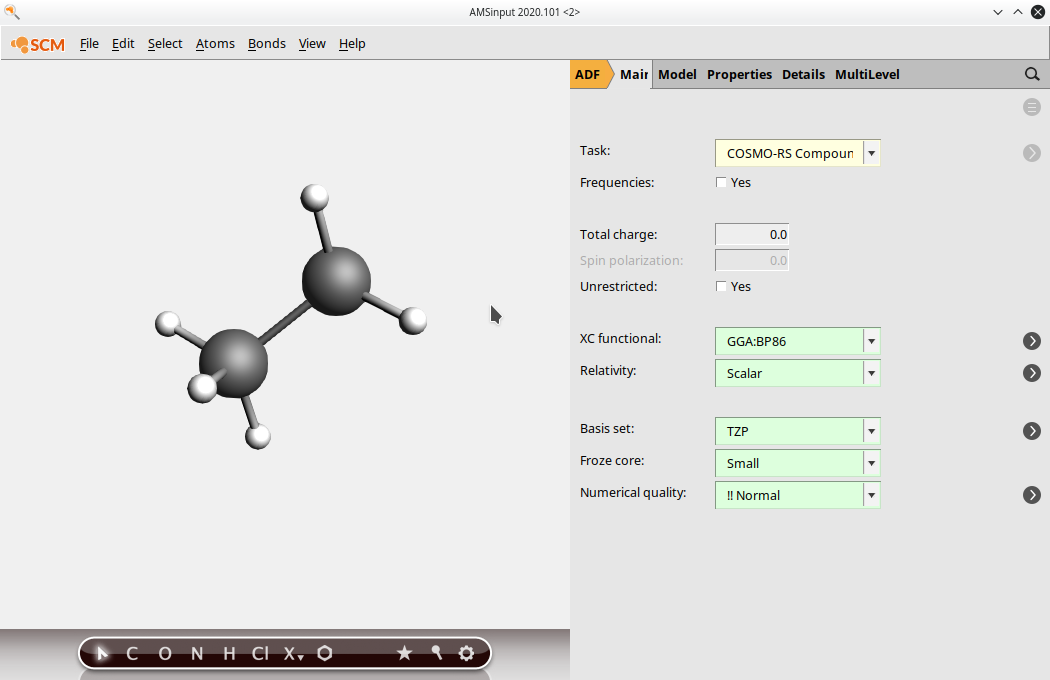
(2) Select the atoms that should be replaced by other monomer units in the full polymer. Click on Atoms → Change Atom Type and replace the atoms with “Xx”. Note: depending on how you have inputted the structure, you may have to add these atoms. Be sure to check that all of the correct atoms are present. Select multiple atoms by holding the Shift key and clicking.
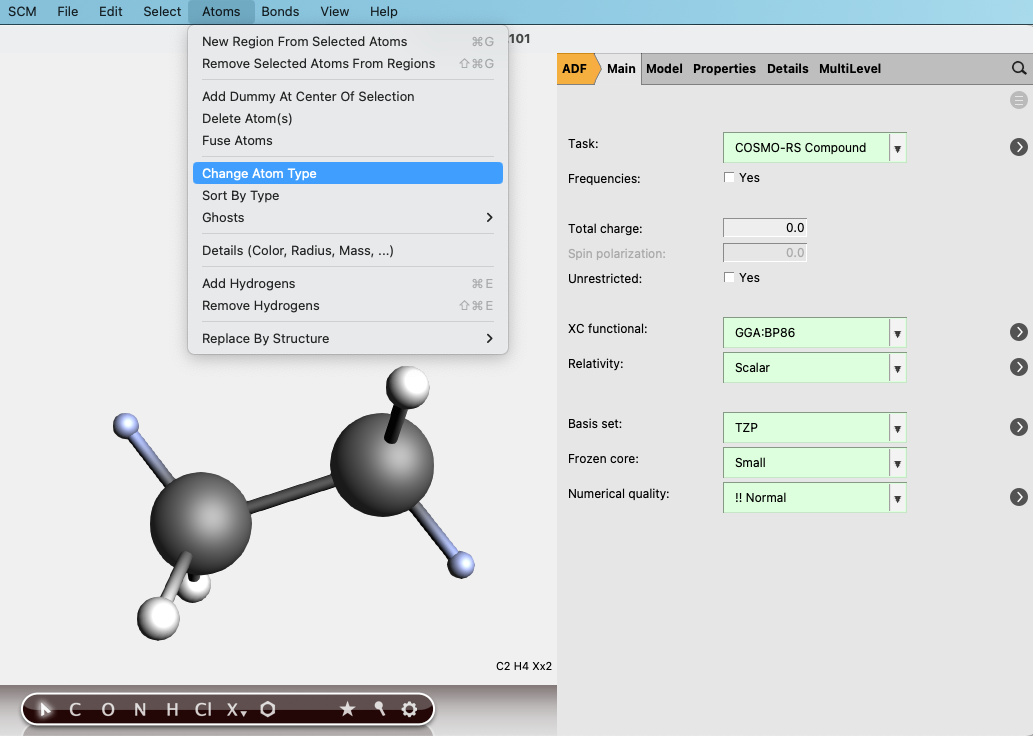
(3) Go to Model → Solvation and click “Generate” next to “COSKF trimer”
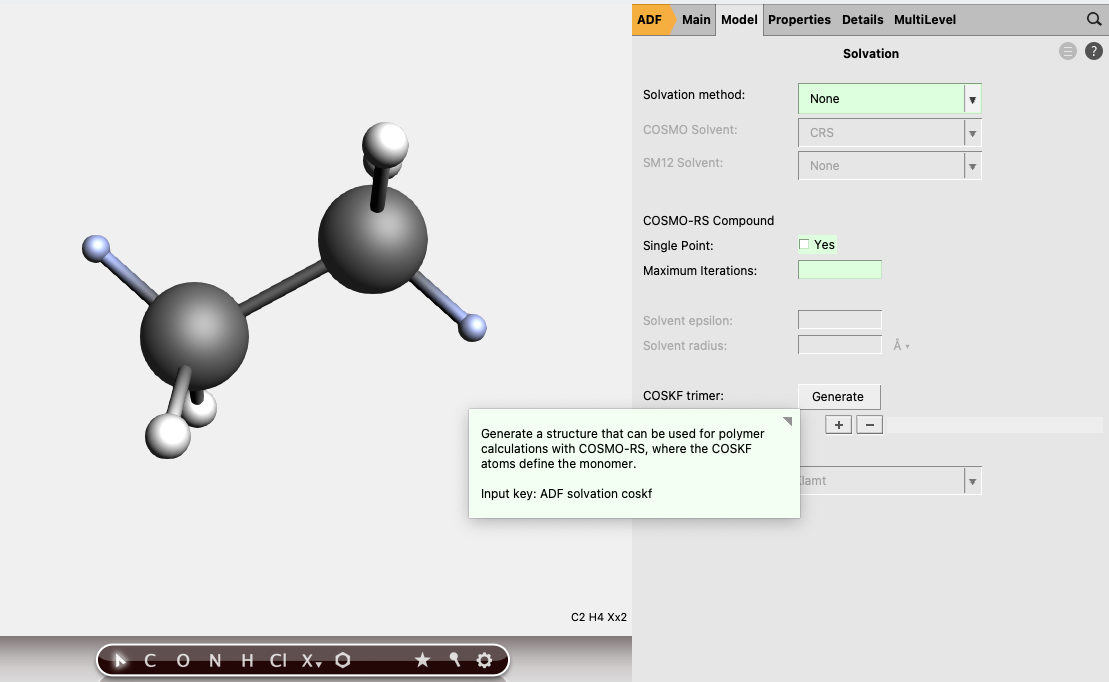
(3.1) (Optional, but recommended) Right click the  and select GFN1-xTB. This will start a pre-optimization with DFTB method and can provide a good initial geometry for the subsequent ADF calculation.
and select GFN1-xTB. This will start a pre-optimization with DFTB method and can provide a good initial geometry for the subsequent ADF calculation.
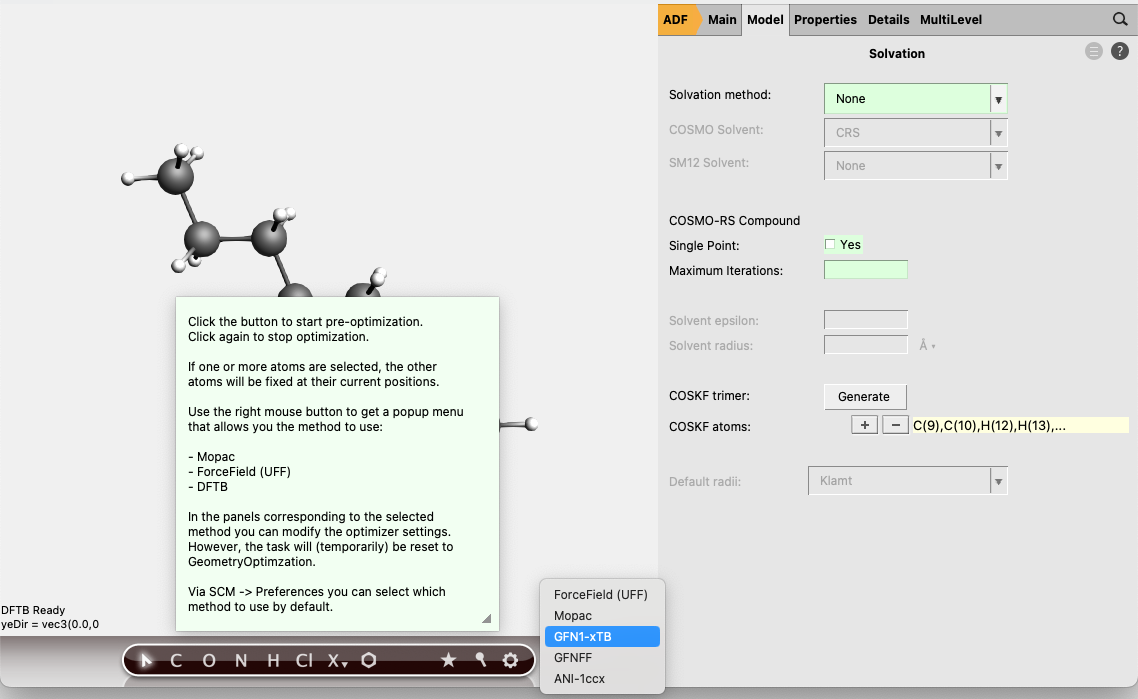
(4) Go back to the ADF panel and select Task: COSMO-RS Compound save your input file
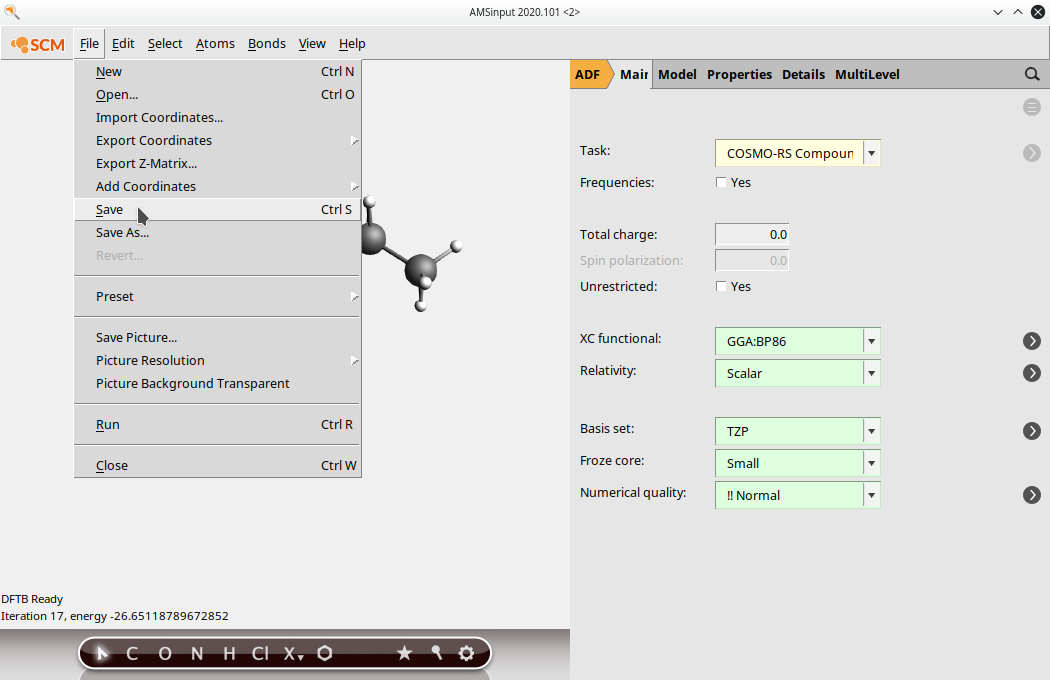
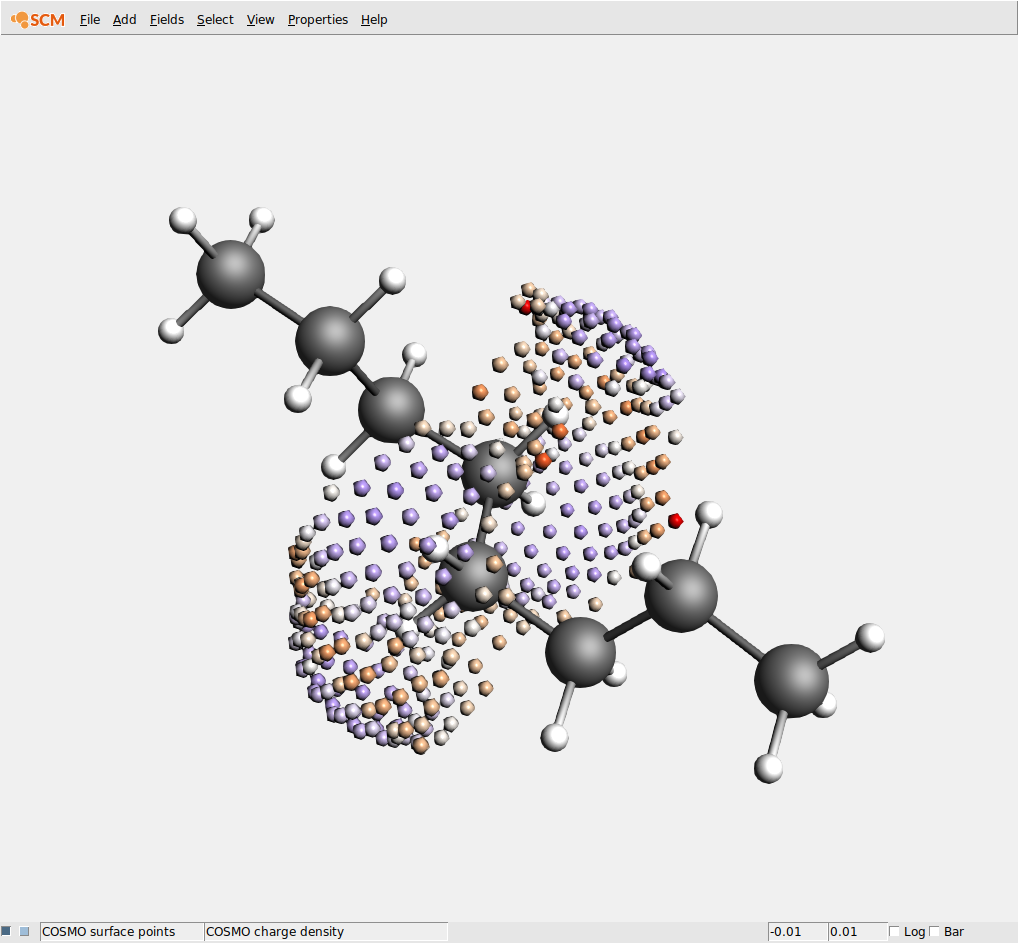
(5) Run the job (File → Run). After running the job, open the newly created .coskf file in the COSMO-RS GUI. Note that depending on the order of opening GUI windows the .coskf file may already have been opened by the COSMOS-RS GUI. You can see the COSMO surface by first selecting your new compound on the Compounds → List of added compounds window. Then press the “Show 3D” button, which opens an AMSview window. From AMSview, click Properties → COSMO: Surface Charge Density → On COSMO surface points. This could produce the image below:
Method 2: Using FastSigma to estimate a polymer’s sigma profile¶
Because ADF calculations for large polymers can be expensive, it is sometimes preferred to estimate the necessary parameters to perform a COSMO-RS calculation. This can be done with the FastSigma tool available from the GUI or the command line. To use this tool from the COSMO-RS GUI, use the following steps:
FS1 in the ModelC{-}C{n+}(c1ccccc1) in the SMILESThe above steps should involve the following (main) step:
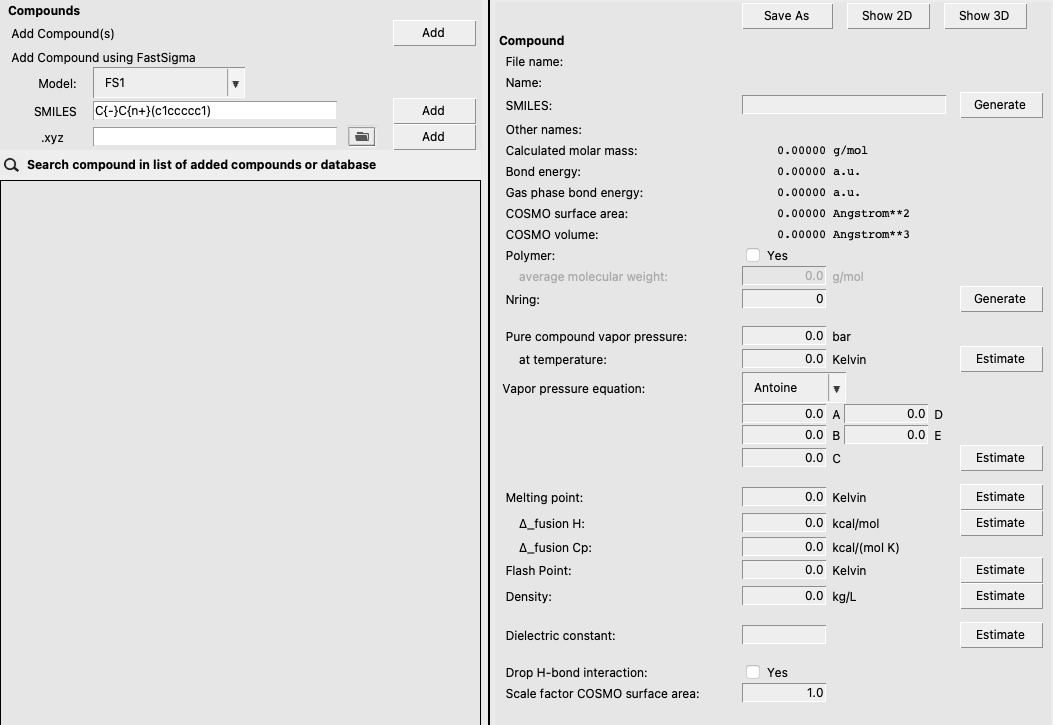
Inputting necessary property values¶
In the rest of the tutorial only polymers from the polymer database will be used.
Polymer/Average Molecular Weight¶
Because polymers of the same type can come in many lengths, it is important to be able to adjust the average molecular weight of a polymer. This is done from the compounds menu: Compounds → List of Added Compounds. Below, we show an example of setting the average molecular weight for Poly(ether_ether_sulfone) (PEES):

Additionally, to specify that a compound should be treated like a polymer, the Polymer box should be checked (shown in the image above). The number of repeat units will be set to the average molecular weight divided by the molar mass of the compound (the compound is assumed to the be monomer). Note that a default value of 10000 g/mol is used if no input is given.
Density¶
Due to a modified combinatorial term for polymers, densities are required for every species in a calculation involving any polymers. In the ADFCRS database, many polymers already have a recommended density value, which should provide reasonable results under standard temperature and pressure conditions. However, it is well-known that for many polymers these density values can be sensitive to temperature and solvent. In these cases – or in cases where there is no recommended density value given – the density can be input from the compounds menu: Compounds → List of Added Compounds.
Here, we show an example of inputting the density for Poly(ether_ether_sulfone) (PEES):

Note
Many polymers in the Polymer Database already have a given experimental density value. This value is automatically used for the calculation if the user does not input a different value for density. These experimental values come from the the Polymer Database Website.
Example polymer calculations¶
Polymers can be used just like other compounds in COSMO-RS/-SAC: they are simply selected as pure components or components of a mixture. If any polymers are present in the system (as indicated by the Polymer flag on the List of Added Compounds menu), then a number of special properties will be calculated. Because polymer calculations are done just like calculations for small molecules, this tutorial will cover a few example calculations without going into the details of every possible type of calculation. For a comprehensive tutorial about different problem types with the COSMO-RS GUI, we redirect the reader here.
Note
To simplify the examples, the molecular weight of the polymer has not been specified. As a result, a default value of 10,000 g/mol will be used to calculate the special properties.
Activity coefficients¶
For this calculation, we estimate the activity coefficients in a Poly(ethylene)/Benzene mixture. Because this is a calculation involving polymers, we will need the density of both Poly(ethylene) and Benzene. There is a recommended value of Poly(ethylene) given (0.852), so we can use that value. For Benzene, we’ll enter a value of 0.876. Use the following summary of steps to perform the calculation:
The result of this calculation is the following:
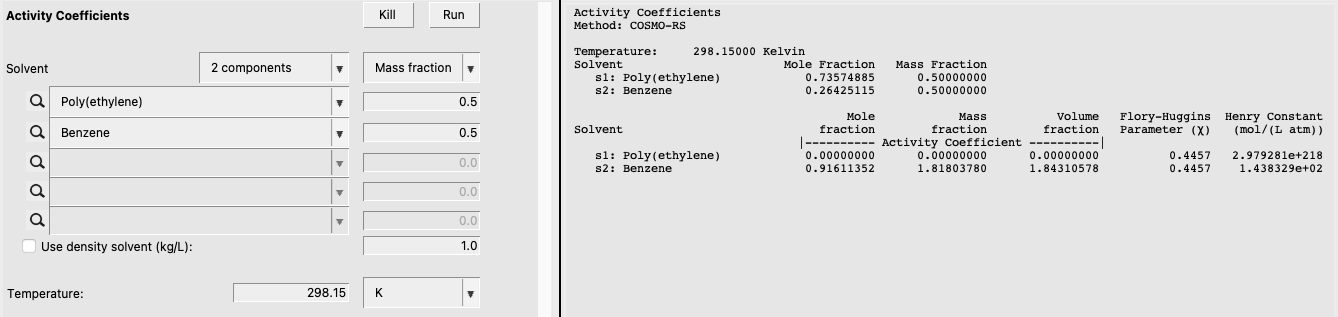
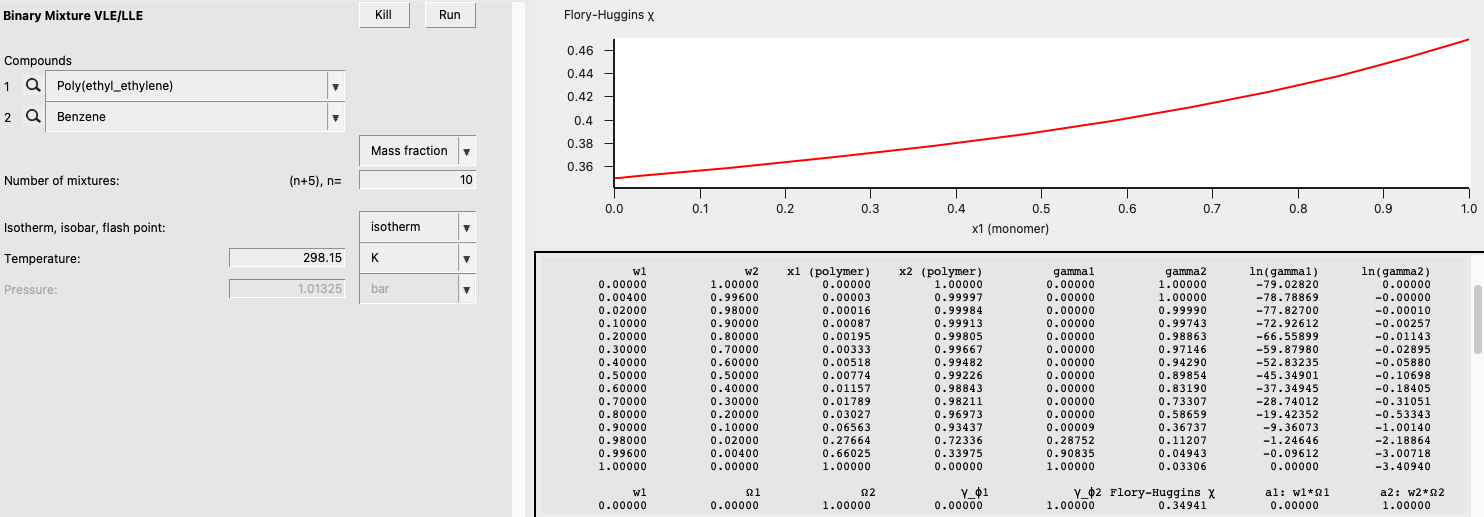
Notice that some non-standard properties are given: weight fraction activity coefficient (= mass fraction activity coefficient), volume fraction activity coefficient, and Flory-Huggins \(\chi\).
Vapor pressure of a mixture¶
Now, we will estimate the vapor pressure of a mixture of Poly(dimethylsiloxane), Methanol, and n-Hexane. Use the following summary of steps to perform the calculation:
The result of this calculation is the following:
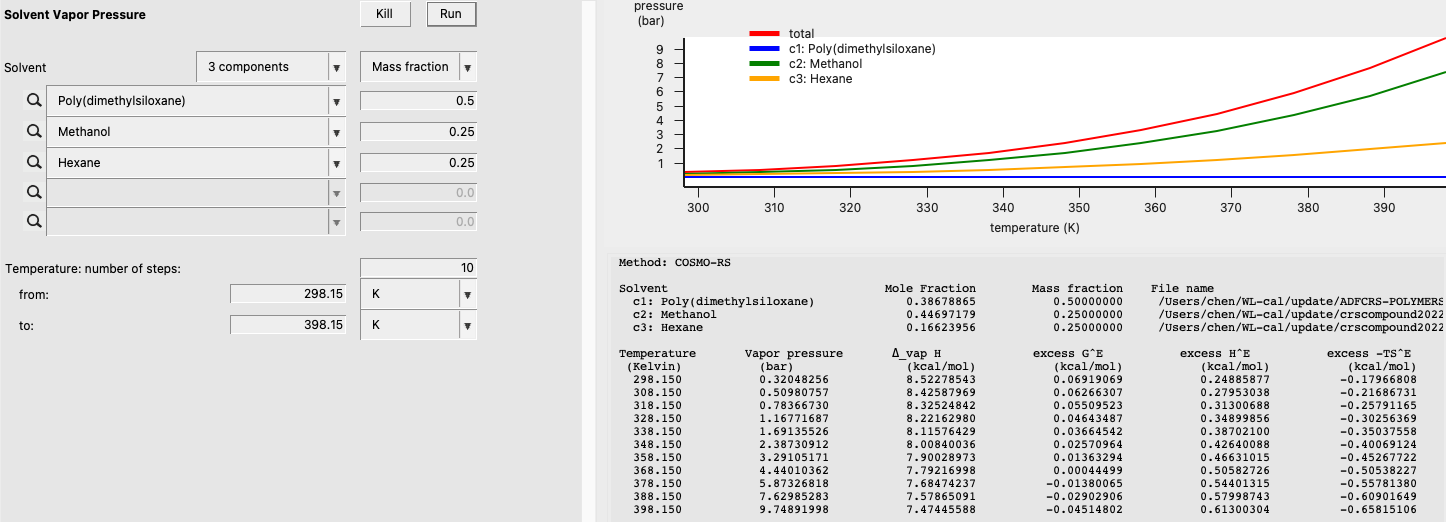
Note that estimated properties are calculated with the Property estimation tools
Partition Coefficients (LogP)¶
Now, we’ll calculate the partition coefficient of Methanol between a water and poly(ethylene) phase.
The output of this calculation is shown below:
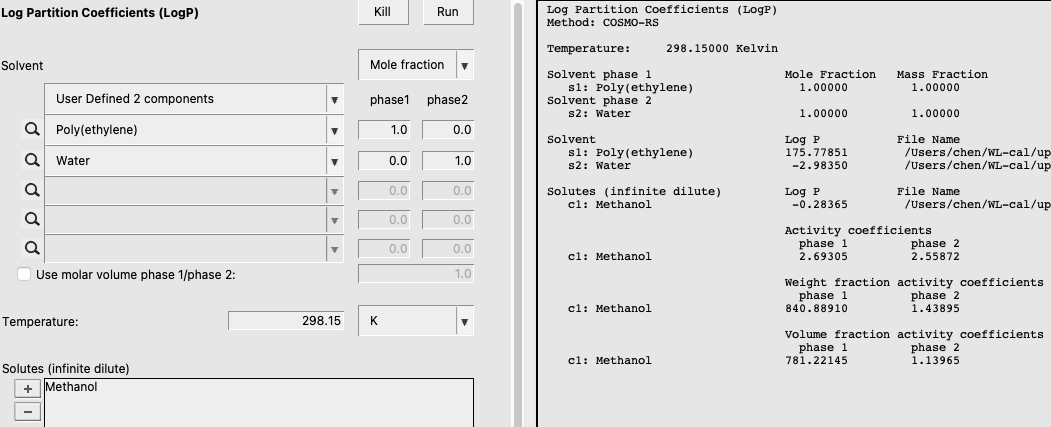
Solubility in Pure Solvents¶
In this example, we’ll calculate the solubility of Hexane gas in Poly(styrene) over the temperature range 398.15 K to 498.15 K and over the pressure range 1.0 bar to 4.0 bar.
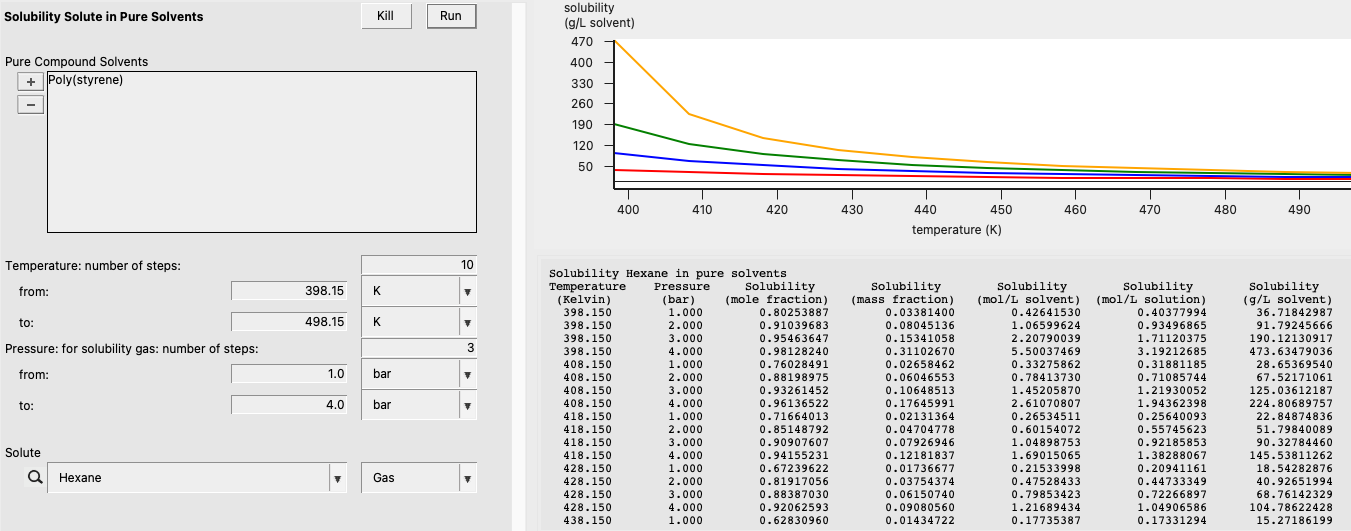
Binary Mixture and Flory-Huggins \(\chi\)¶
In this example, we will calculate binary mixture properties (including the \(\chi\)) over the entire range of compositions. We will do this for a binary mixture of Benzene and Poly(ethyl_ethylene) and use COSMO-SAC (the 2013-ADF Xiong parameters).
The result of this calculation is the following: